Xps peak pure sulphur s2p
XAS data analysis suggested a square-pyramidal geometry around the transition metal with H2S in the apical position for the pentacoordinated platinum units. Together with other authors, we attribute the S2p 3/2 peak at 162.2 eV to sulphur atoms bound to gold, since a shift of the order of 23 eV towards lower binding energies with respect to the pure sulphur state (E B 164.0 eV) is typical of sulphur metallic compounds, such as CuS and FeS. The user typically observes either splitting between components or asymmetric peak shape due. Effect of spin-orbit splitting can be observed for sulfur elements and compounds. XPS measurements evidenced the presence of S in the polymetallayne samples exposed to H2S, and the measured S2p binding energy values correlate with H2S adsorbed by means of sulfur atoms chemically bonded to metal atoms, owing to the formation of sulfur-containing adducts. S2p peak has closely spaced spin-orbit components (1.16eV, intensity ratio0.511). The polymetallayne trans-, the tetranuclear cyclic oligomer cis-4, were exposed to hydrogen sulfide and then investigated by X-ray photoelectron (XPS) and X-ray absorption (XAS) spectroscopies, in order to shed light on the gas/polymer interaction associated to the sensing properties of these materials.
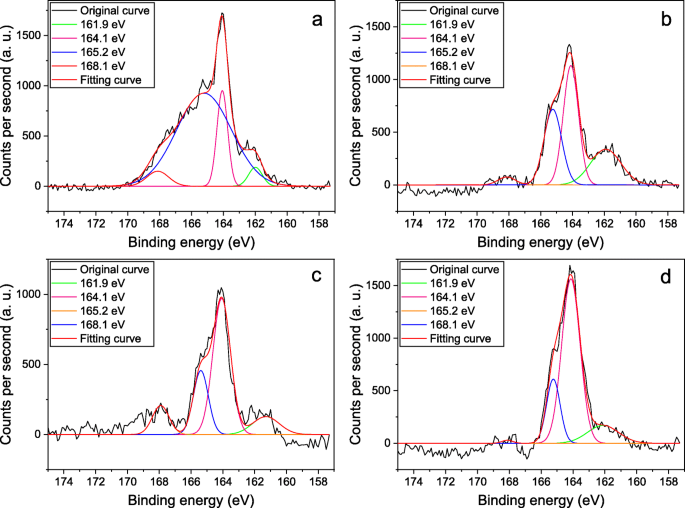
The interaction between gaseous H2S and the surface of several metal-containing oligomers, investigated by emission and absorption spectroscopies, is presented and discussed.
